

MeticTouch®
Non-invasive Ultrasound Ocular Drug Delivery Platform


Novel ultrasound
drug induction system
Compatible to
wide range of APIs
Profiles proven in
preclinical trials
Patent portfolio in
13 major countries
Demonstrated efficacy
and bioavailability
with multiple drug products

Problems of Traditional Eye injections:

Solutions provided by MeticTouch®:
Ultrasound treatment has a shorten total patient involved time, compared to traditional eye injection.
| TRADITIONAL EYE INJECTION | ULTRASOUND TREATMENT |
| PRE-OPERATION ACTIVITIES |
|---|
1. Seat patient and brief 
2. Apply anesthetic and disinfectant drops 
3. Covered and secured region of interest with drape  |
1. Turn on device and select from pre-set treatment protocols 
2. Prepare single-use drug applicator and attached to device *All pre-operation activities in ultrasound treatment only involve HCPs. |
| OPERATION ACTIVITIES |
|---|
1. Place speculum onto patient eyes 
2. Apply local anesthetic with swap 3. Mark injection site (3.5mm from limbus) 4. Instruct patient to look to one side and keep eye/head still 5. Inject into patient's eye 
6. Hold cotton bud at the injection site  |
1. Seat patient and brief 
2. Apply disinfectant drops 
3. Instruct patient to look to one side 4. Apply ultrasound drug induction for 60 secs 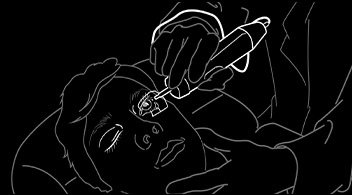 |
| POST-OPERATION ACTIVITIES |
|---|
1. Apply antibiotics eye drops 2. Remove drape and speculum 3. Debrief  |
1. Debrief  |
| TOTAL PATIENT INVOLVED TIME | |
|---|---|
| 10 mins | 5 mins * Treatment procedure subject to regulatory approval. |
MeticTouch® approaches the eye

With low-frequency ultrasound, changes in acoustic pressure creates oscillating microbubbles that contracts and expands
The oscillation causes movement in the surrounding liquid and form microstreaming by shearing force

This eventually creates non-permanent micropores in the cell layer, allowing therapeutics to enter the eye, through the sclera and directly to the retina.
Our advanced technology is highly compatible to various drug products, targeted to be applicable to a wide range of ocular diseases management, in particular to posterior diseases where injection is usually required for treatment.


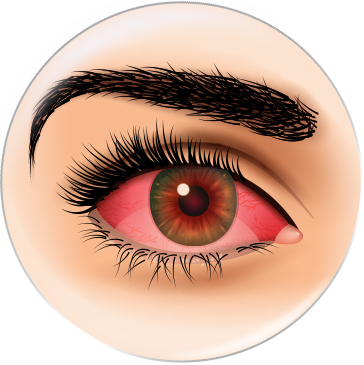
It is a common condition that affects the middle part of your vision. It usually first affects people in their 50s and 60s. It can make everyday activities like reading and recognising faces difficult. Without treatment, your vision may get worse. This can happen gradually over several years ("dry AMD"), or quickly over a few weeks or months ("wet AMD").The exact cause is unknown. It's been linked to smoking, high blood pressure, being overweight and having a family history of AMD.2
Diabetic Macular Edema (DME) is a severe complication of Diabetic retinopathy (DR) that occurs specifically as a result of inadequately treated diabetes mellitus (DM), has overtaken proliferative diabetic retinopathy (DR) as the most common cause of vision impairment in individuals with diabetes mellitus (DM). In recent epidemiologic studies, approximately 30% of patients worldwide with diabetes mellitus (DM) were found to have vision-threatening Diabetic retinopathy (DR); and in the United States, 3.8% of patients were found to have DME.3
Is a serious, sight-threatening intraocular inflammatory condition characterized by inflammation of the uvea including iris, ciliary body, and choroid.4
* Simulated vision for reference only.

MeticTouch® is proven in compatibility with major and common treatment molecules.
Animal studies indicating no abnormality
after ultrasound treatment up to 6 months

No obvious changes in retina thickness (OCT)
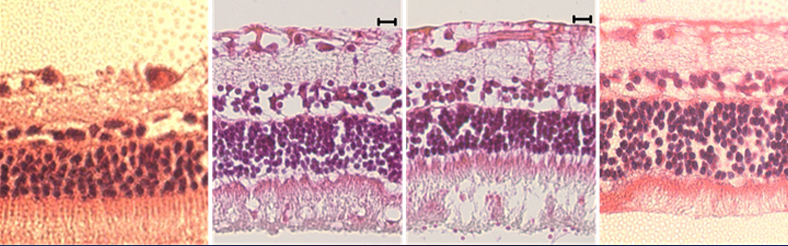
Histology indicate fully intact retina without any abnormality
References